Lithium carbonate by Behansar Co.

Lithium carbonate (Li₂CO₃) is an inorganic compound classified among lithium salts. It appears as a white, odorless solid and represents one of the most established and widely utilized pharmaceutical forms of elemental lithium. Clinically, lithium carbonate plays a pivotal role in the management of psychiatric disorders—most notably bipolar disorder. Within pharmaceutical practice, it is recognized as a primary mood stabilizer and remains a cornerstone agent in evidence-based psychiatric treatment protocols.
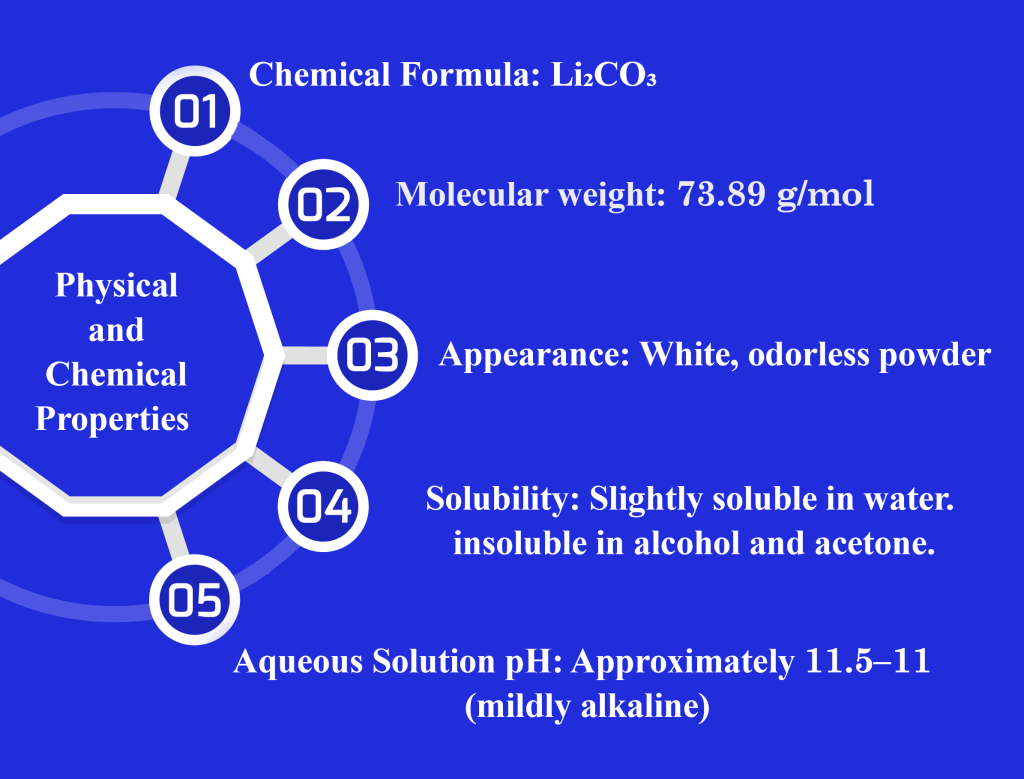
Its stable crystalline structure ensures high physicochemical stability under ambient conditions, making lithium carbonate particularly suitable for solid dosage form manufacturing, including tablets, capsules, and extended-release formulations.
Pharmaceutical-Grade Requirements for Lithium Carbonate
Lithium carbonate intended for pharmaceutical applications must meet stringent quality specifications, including:
- Minimum purity of 99%
- Absence of heavy metal impurities and undesirable contaminants
- Controlled and uniform particle size distribution to ensure consistent performance during formulation and manufacturing processes
- Full compliance with recognized pharmacopeial standards, such as the United States Pharmacopeia (USP), European Pharmacopoeia (EP), or British Pharmacopoeia (BP)
These criteria are critical to ensuring the safety, therapeutic efficacy, and stability of lithium-containing medicinal products. Careful selection of pharmaceutical-grade active ingredients is therefore essential in drug manufacturing.
Common Dosage Forms in Iran
- 300 mg immediate-release tablets
- 400 mg extended-release tablets, designed for patients requiring more stable serum lithium level control
Lithium carbonate serves as the key active pharmaceutical ingredient (API) in mood-stabilizing medications. Currently, several pharmaceutical manufacturers with varying formulations contribute to meeting the country’s therapeutic demand. Behan Sar Company is a leading manufacturer of BP/USP-grade lithium carbonate, operating advanced production lines in compliance with Good Manufacturing Practice (GMP) standards. In addition to high-quality supply, the company provides expert technical consultation in excipient selection and delivers comprehensive documentation and analytical data to support formulation development and manufacturing processes. Through continuous supply and regulatory-compliant production, Behan Sar plays a significant role in sustaining the national pharmaceutical market for lithium carbonate.
Product Information
Product Number | Product Name | Pharmacopoeia | Product Features | Storage Conditions |
151300153 | Lithium Carbonate | USP | Sparingly soluble in water. very slightly soluble in alcohol. | Preserve in well-closed containers. |

